Atomic Theory
There were many theories by scientist about the atom and how it worked and the parts it had to it. The first scientist was Democritus. His theory of the atom was that it could not be destroyed and atoms of the matter would stay the same forever. The John Dalton, another scientist, did not think all matter was made up of atoms. Then a scientist Thomson Plum gave the first impression that atoms were made up of small parts. He made a Model that showed that an atom has a negative and a positive charge. Later in 1866, Goldstein gave a name to these charges, proton being the positive charge and electron being the negative charge. Then a scientist James Chadwick said there was a part that had no charge, called neutron. Rutherford Gold studied the atom in 1908 and discovered that the atom had a nucleus in the middle which was positive, and everything else on the outside was negative. Then in 1913 Bohr discovered that electron around the nucleus orbit the nucleus.
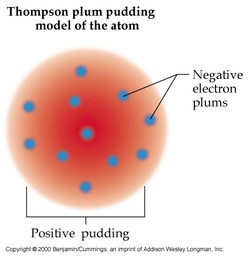
Thomas Plum's Model
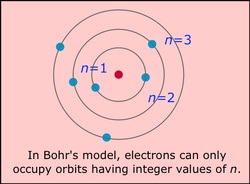
Bohr's Model
