Reaction Rates
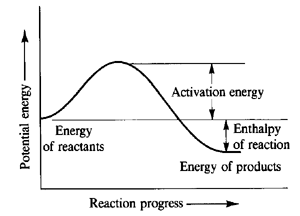
A reaction rate is to see how a chemical equation has progress over a period of time. They can see if a chemical equation is fast or slow, the faster a chemical equation the more likely it will explode and the slower a chemical reaction the more time it takes to destroy something. By seeing how slow or fast a chemical reaction is you can see the more energy is released or is taken in. During a chemical reaction particles collide with each other, and the more they collide there more the reaction is increased. If particles collide slower they are decreased. Reaction rates are also affected by Temperature, surface area, concentration, stirring and catalyst. Reaction rates increase the higher the temperature is, the lower it is decreases more. The surface area will increase when the particles decrease. The more reaction is stirred moves the particles around more, speeding up the reaction. Concentration is the number of particles a reaction has. So an increase in concentration speed up a reaction and a decrease in concentration slows it down. Catalysts speed up the reaction with chemically affecting the reaction.