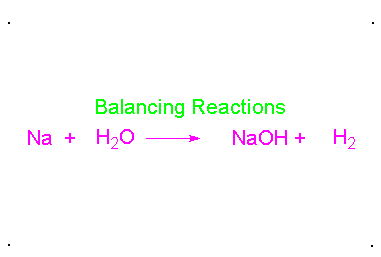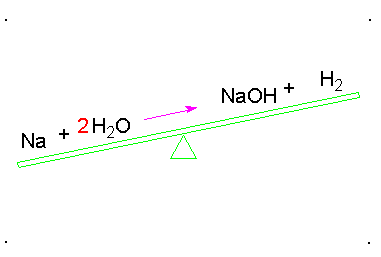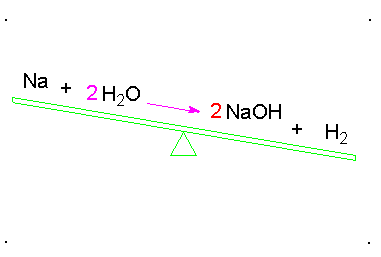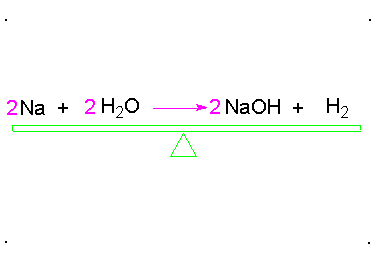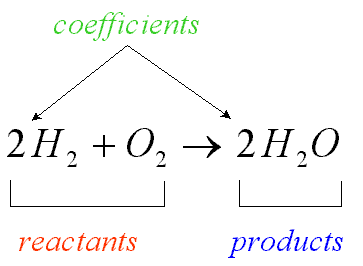Balancing chemical equations
When a chemical equation is present, the elements must have the same number on both sides. To do this a coefficients must be added to the element. A coefficient is the number that goes in front of the element or compound so that it matches the other side of the equation. Here is an example:
K + H2 = KH
The K does not start with any electrons so it stays the same, The H has a 2 because it is diatomic and that means that the element always has 2 electrons when it is by itself. and it creates KH because neither element has an electron to give to the other. Now that it is set up, we can balance the equation
There is 1 K and 2 hydrogen on the left side (reactant side) and 1 K and H on the left side (product side) so we have to give a coefficient 2 to K on the left side and a 2 to the KH. (2x1=2)
2K + H2 = 2KH
Now there are 2 K on both sides and 2 H on both sides. And the equation is balanced.
K + H2 = KH
The K does not start with any electrons so it stays the same, The H has a 2 because it is diatomic and that means that the element always has 2 electrons when it is by itself. and it creates KH because neither element has an electron to give to the other. Now that it is set up, we can balance the equation
There is 1 K and 2 hydrogen on the left side (reactant side) and 1 K and H on the left side (product side) so we have to give a coefficient 2 to K on the left side and a 2 to the KH. (2x1=2)
2K + H2 = 2KH
Now there are 2 K on both sides and 2 H on both sides. And the equation is balanced.