Classifying Matter
The Classification of matter include its main substances: Solids, Liquids and Gases
Solids: A Solid never loses it's shape, it will always stay the same. Like a rock, a rock is a solid and will always be a solid even if it breaks in half. Solids always keep the same shape, and are not compressible. The atoms in a solid are very compacted together and do not move. Examples of a solid: Sand, Ice, Rocks, Table, Wood
Liquids: It.s water, blood, soda, olive oil. Liquid is anything that can fill up a container. The atoms inside a liquid are loose and move just a little bit, but do not spread out from each other. Examples: Water, Blood, Milk, Syrup
Gases: Are all around us. They are in the air and they are what fill up a balloon. The atoms in a gas are very spread out and are constantly moving. They fly all throughout the air and are constantly bouncing off each other. Example: Air, Helium, Steam, Water Vapor
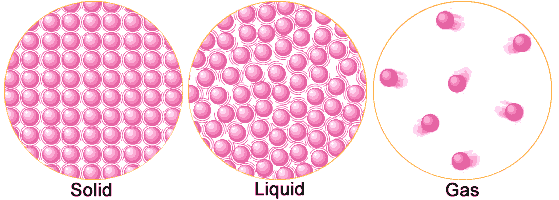
Atoms in a Solid, Liquid, and Gas
Solids: A Solid never loses it's shape, it will always stay the same. Like a rock, a rock is a solid and will always be a solid even if it breaks in half. Solids always keep the same shape, and are not compressible. The atoms in a solid are very compacted together and do not move. Examples of a solid: Sand, Ice, Rocks, Table, Wood
Liquids: It.s water, blood, soda, olive oil. Liquid is anything that can fill up a container. The atoms inside a liquid are loose and move just a little bit, but do not spread out from each other. Examples: Water, Blood, Milk, Syrup
Gases: Are all around us. They are in the air and they are what fill up a balloon. The atoms in a gas are very spread out and are constantly moving. They fly all throughout the air and are constantly bouncing off each other. Example: Air, Helium, Steam, Water Vapor
Atoms in a Solid, Liquid, and Gas