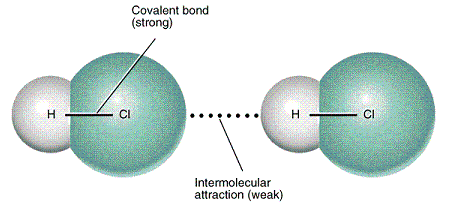
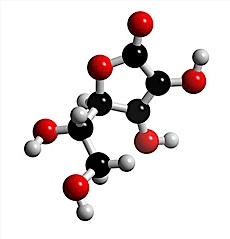
Chemical bonding
Chemical bonding is combining of atoms into molecules, ions, and crystal. When atoms
get in contact with each other, their electrons interact and tend to
distribute themselves to what they need to be bonded as and get their energy how they want it. If the total energy of atoms is lower than the other atoms, they
then bond together.
There were few ideas that helped make the come up of chemical bonding. It Came during early in the 20th century. Many of the the odels that were made can explain the chemical bonding.
A key concept in a discussion of chemical bonding is that of the molecule. Molecules are the smallest units of compounds that can exist. One feature of molecules that can be predicted with reasonable success is their shape. Molecular shapes are of considerable importance for understanding the reactions that compounds can undergo, and so the link between chemical bonding and chemical reactivity is discussed briefly in this article.
There were few ideas that helped make the come up of chemical bonding. It Came during early in the 20th century. Many of the the odels that were made can explain the chemical bonding.
A key concept in a discussion of chemical bonding is that of the molecule. Molecules are the smallest units of compounds that can exist. One feature of molecules that can be predicted with reasonable success is their shape. Molecular shapes are of considerable importance for understanding the reactions that compounds can undergo, and so the link between chemical bonding and chemical reactivity is discussed briefly in this article.