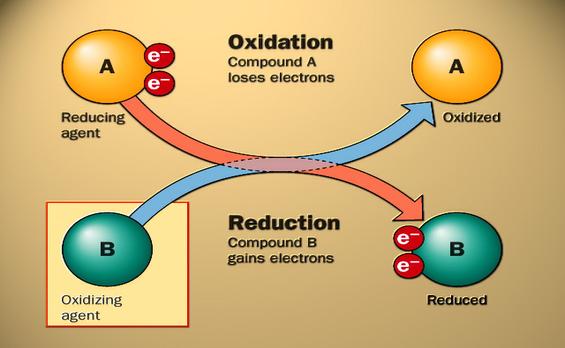
oxidation reduction reactants
A reduction is when an element gains an 1 or more electrons. Oxidation is when and element loses electrons. A redox is when a oxygen is added to a chemical reaction. A reduction and an oxidation cannot happed without one happening to and the other not. Because when an oxidation occurs, another element is being reduced and when a element goes threw reduction other element is being oxidized. With these reactions taking place they have oxidation numbers