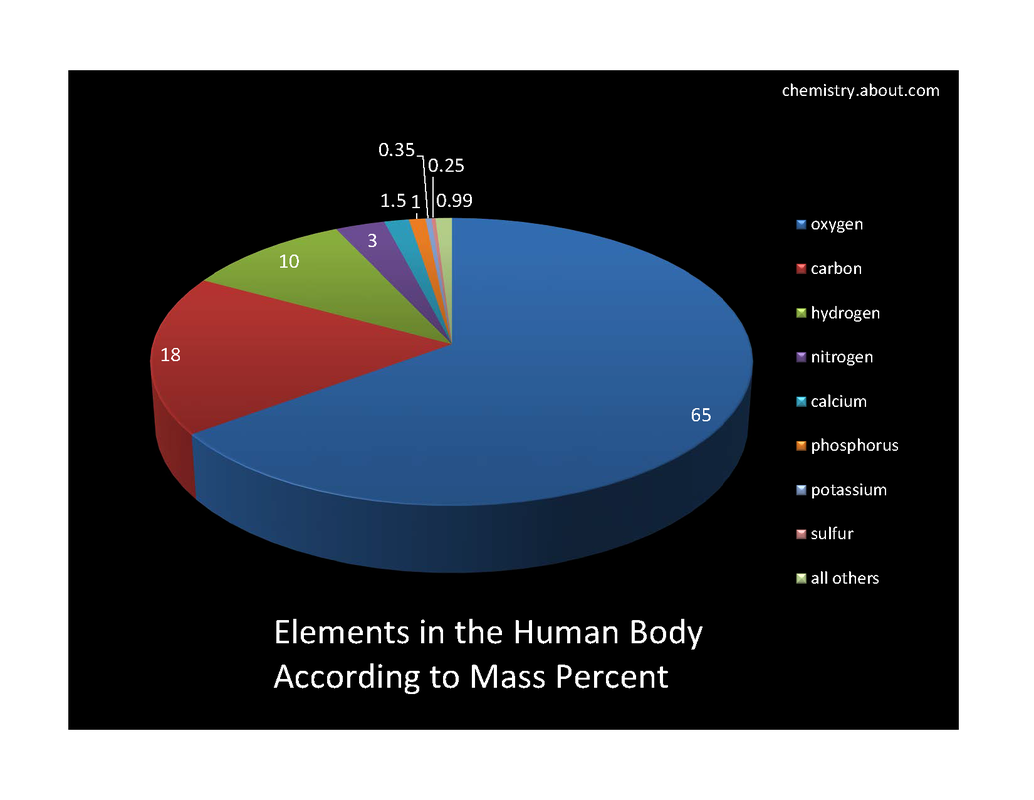
percent composition
Percent composition is the percent of total mass a volume something has. It will refer to weight percent or volume percent. to find the percent comp of something, there is an equation that will help find it:
First you have to find the atomic masses of the elements: use for example: Na, H, C, and O
Na is 22.99
H is 1.01
C is 12.01
O is 16.00
Next, determine how many grams of each element are present in one mole of NaHCO3:
22.99 g (1 mol) of Na
1.01 g (1 mol) of H
12.01 g (1 mol) of C
48.00 g (3 mole x 16.00 gram per mole) of O
Then you add up all the elements to find The mass of one mole of NaHCO3 is:
22.99 g + 1.01 g + 12.01 g + 48.00 g = 84.01 g
And the mass percentages of the elements are
Na = 22.99 g / 84.01 g x 100 = 27.36 %
H = 1.01 g / 84.01 g x 100 = 1.20 %
C = 12.01 g / 84.01 g x 100 = 14.30 %
O = 48.00 g / 84.01 g x 100 = 57.14 %
First you have to find the atomic masses of the elements: use for example: Na, H, C, and O
Na is 22.99
H is 1.01
C is 12.01
O is 16.00
Next, determine how many grams of each element are present in one mole of NaHCO3:
22.99 g (1 mol) of Na
1.01 g (1 mol) of H
12.01 g (1 mol) of C
48.00 g (3 mole x 16.00 gram per mole) of O
Then you add up all the elements to find The mass of one mole of NaHCO3 is:
22.99 g + 1.01 g + 12.01 g + 48.00 g = 84.01 g
And the mass percentages of the elements are
Na = 22.99 g / 84.01 g x 100 = 27.36 %
H = 1.01 g / 84.01 g x 100 = 1.20 %
C = 12.01 g / 84.01 g x 100 = 14.30 %
O = 48.00 g / 84.01 g x 100 = 57.14 %