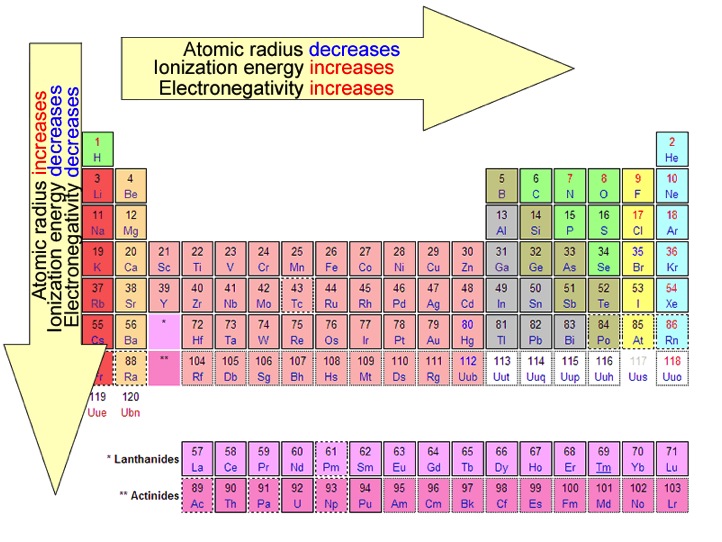
Periodic trends
Periodic Trends are different ways to tell the certain size and its properties. The main types of periodic trends that are used are electron Affinity, electronegativity, Atomic Radius, and Ionization Energy. These groups help because of the similarities in the groups and periods that exist and the nature of the elements.
Electron Affinity- This is when an atom wants to accept an electron. Its trend is that is increase in a period (from right to left) and decreases in a group (up to down)
Electronegativity- This is an atoms ability to bind with other electrons. Its trend is that it increases in a period (from right to left) and decreases in a group (up to down)
Atomic Radius- This is to tell how big an atom is. Its trend is Decrease across a period(left to right) and increase up a group (down to up)
Ionization Energy- When a electron needs to be removed from an atom at a certain energy. The trend is to increases in a period (from right to left) and decreases in a group (up to down)
Electron Affinity- This is when an atom wants to accept an electron. Its trend is that is increase in a period (from right to left) and decreases in a group (up to down)
Electronegativity- This is an atoms ability to bind with other electrons. Its trend is that it increases in a period (from right to left) and decreases in a group (up to down)
Atomic Radius- This is to tell how big an atom is. Its trend is Decrease across a period(left to right) and increase up a group (down to up)
Ionization Energy- When a electron needs to be removed from an atom at a certain energy. The trend is to increases in a period (from right to left) and decreases in a group (up to down)