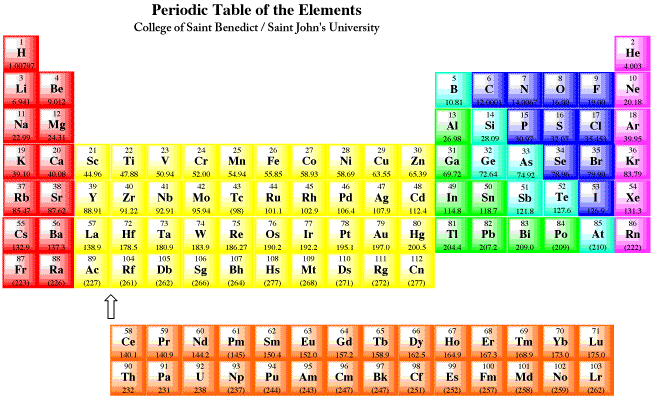
Periodic table
On the periodic table, there are many elements and they are put into groups and periods. A group on the periodic table are elements that are in the same column that go up and down. A period of elements are in the same row that go from left to right. Each element has a certain amount of valence electrons hat fit into their groups. A valance electron is are electrons that are on the outer energy level. The goal for each element is to either have 0 VE or 8 VE. Each group has a name:
Group 1 is called the Alkali Metals. All the elements in this group have 1 VE and are the most reactive of the metals. They react strongly with non metals and water. Since they are so reactive they cant be found in nature and have a silvery appearance to them.
Group 2 are called the Alkaline Earth Metals. All these elements have 2 VE. These elements are much harder than group 1. They are very reactive but not as much as group 1. Like group 1 they cant be found in nature. The special part about this group is that many of the elements are found in the earths crust.
Groups 3-12 are called transition Metals. Most of the elements in these groups have 2 VE and are not as reactive as groups 1 and 2. Some of them are found in nature like Gold, Copper, and Silver.
Group 17 is called the Halogens. They all have 7 VE and are the most reactive non-Metals and contains the most reactive element, Fluorine.
Group 18 is called the Noble Gases. They have 8 VE, except for helium that only has 2. Are weakest in reactions because they already have 8 VE.
Group 1 is called the Alkali Metals. All the elements in this group have 1 VE and are the most reactive of the metals. They react strongly with non metals and water. Since they are so reactive they cant be found in nature and have a silvery appearance to them.
Group 2 are called the Alkaline Earth Metals. All these elements have 2 VE. These elements are much harder than group 1. They are very reactive but not as much as group 1. Like group 1 they cant be found in nature. The special part about this group is that many of the elements are found in the earths crust.
Groups 3-12 are called transition Metals. Most of the elements in these groups have 2 VE and are not as reactive as groups 1 and 2. Some of them are found in nature like Gold, Copper, and Silver.
Group 17 is called the Halogens. They all have 7 VE and are the most reactive non-Metals and contains the most reactive element, Fluorine.
Group 18 is called the Noble Gases. They have 8 VE, except for helium that only has 2. Are weakest in reactions because they already have 8 VE.