bond polarity
Bond polarity is when atoms are unequally shared between atoms that have been bonded together that end in a positive and negative.
Electrons are not always shared equally between two bonding atoms; one atom might have more force on the electron cloud than the other. This unequal sharing of electrons may crate an electric dipole: which is a separation of positive and negative electric charges.
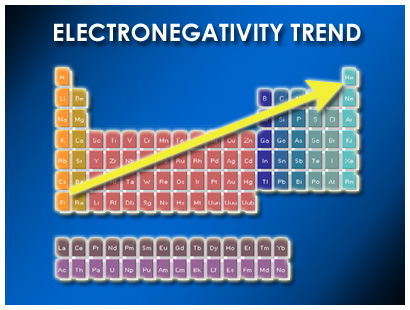
To determine the electron sharing between two atoms, a table of electronegativities can be used to determine which atom will attract more of the electron density. The more electronegative atom will have a greater negative charge around it.
Bonds can fall between in two ways, either from completely nonpolar or to polar. Nonpolar bond occurs when the electronegativity values are the same as each other and have a difference of zero. A polar bond is more correctly of ionic bonding.
Electrons are not always shared equally between two bonding atoms; one atom might have more force on the electron cloud than the other. This unequal sharing of electrons may crate an electric dipole: which is a separation of positive and negative electric charges.
To determine the electron sharing between two atoms, a table of electronegativities can be used to determine which atom will attract more of the electron density. The more electronegative atom will have a greater negative charge around it.
Bonds can fall between in two ways, either from completely nonpolar or to polar. Nonpolar bond occurs when the electronegativity values are the same as each other and have a difference of zero. A polar bond is more correctly of ionic bonding.