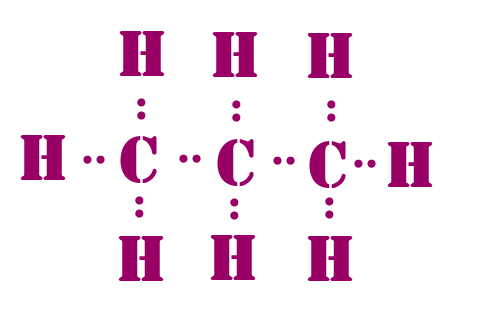Lewis dot structure
The Lewis dot structure is to describe the bonds between atoms of a molecule. The Lewis dot structure can be used for any covalent molecule or coordination compound. The Lewis structure is a simple and easy to use. Atoms are written as their element symbol. Lines are drawn between atoms to describe the chemical bonds. Single bond as one line, a double line has two lines, and a triple bond has three line. Dots are drawn next to atoms to show unbonded electrons. A pair of dots is a pair of excess electrons. Here are steps to making a dot structure. Important note: Every element either needs 0 or 8 electrons.
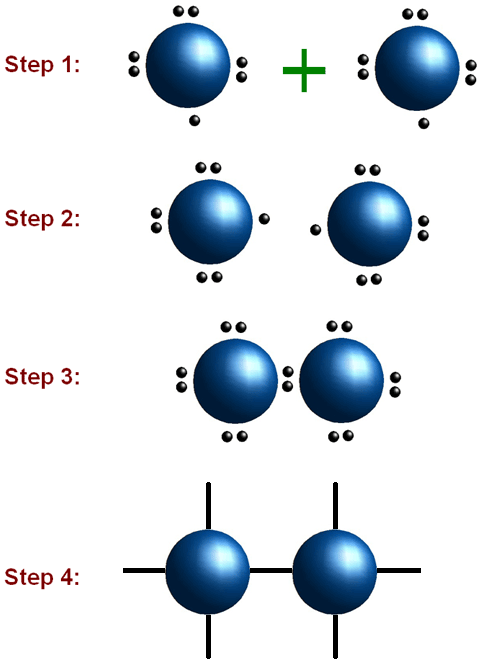
Step 1- Pick a central atom. Once you have selected the central atom, write it down and connect the other atoms to it with a single bond. You may change these bonds to become double or triple bonds if needed.
Step 2- Count the electrons. All the atoms show their valence electrons. This is to determine where the electrons need to go.
Step 3- Now that you have determined how many electrons are needed to each atom, place them on the structure. Start by placing one pair of dots for each pair of valence electrons. Once the lone pairs are placed, it may be necessary to draw a double or triple bond. Now the elements should contain either 0 or 8 electrons.
Step 1- Pick a central atom. Once you have selected the central atom, write it down and connect the other atoms to it with a single bond. You may change these bonds to become double or triple bonds if needed.
Step 2- Count the electrons. All the atoms show their valence electrons. This is to determine where the electrons need to go.
Step 3- Now that you have determined how many electrons are needed to each atom, place them on the structure. Start by placing one pair of dots for each pair of valence electrons. Once the lone pairs are placed, it may be necessary to draw a double or triple bond. Now the elements should contain either 0 or 8 electrons.