history of the periodic table
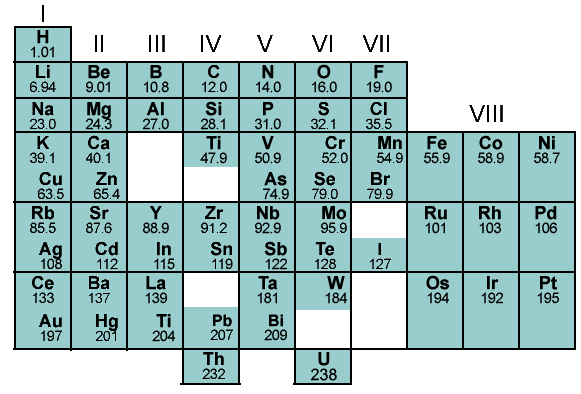
History: In 1860 there were only 60 elements. All of the elements were published into a book and was given an atomic number, properties, and a symbol. While publishing the book, a Russian chemist, Dimitri Mendeleev, came up with a way to organize all the elements. He began to organize them in increasing atomic mass, he realized that his table was periodic by having every 8th element being unreactive. He noticed that there was a problem with the elements Iodine and Tellurium. If they are put in the order of increasing mass, then their protons don't match with their columns. His thinking was that they must have been recorded incorrectly so he switched them. On his table he left a few blank spaces for where he believed undiscovered elements would go. A chemist named Henry Moseley later helped fix the problem with Iodine and tellurium. His idea was to put the elements in increasing atomic number and he created the periodic Law. This Law states that the fit in the order that in their columns they have certain characteristic that match with each other.
Henery Moseley
Henery Moseley