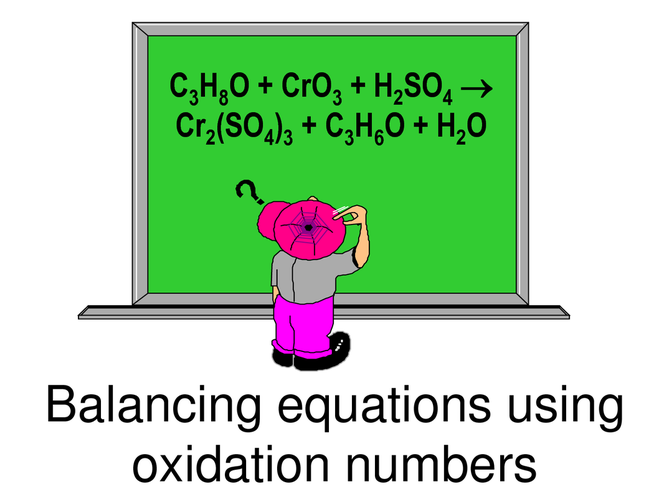
Oxidation numbers equation
Example:
KClO4
If you see on rule 3, the oxidation number of K is +1. IF you look at rule 6, the oxidation number of O is -2 and the total for 4 O atoms is -8. For these two elements ,the total is +1 - 8 = -7. The oxidation number of Cl must be +7 to give a total of zero for all atoms in this formula unit.
KClO4
If you see on rule 3, the oxidation number of K is +1. IF you look at rule 6, the oxidation number of O is -2 and the total for 4 O atoms is -8. For these two elements ,the total is +1 - 8 = -7. The oxidation number of Cl must be +7 to give a total of zero for all atoms in this formula unit.